How the MTHFR gene mutation Affects fertility and pregnancy
One of the most common things I hear is the utter confusion many people find themselves in after they:
- Just discovered the MTHFR gene mutation and fell down an internet rabbit hole try to research and understand it all
- Were tested for it but received no explanation on its effects
- Were prescribed treatments that instead of helping made them feel worse
- Were told it may be one of the facets playing a role in their present health issues, but have no idea what it is
With the wealth of scientific (and at times confusing and complex) information found online surrounding MTHFR, it is no wonder many end up going around in circles and getting confused!
This is can be especially amplified in the case of those searching for clear and reliable information when it comes to the MTHFR gene mutation effects on fertility/preconception and pregnancy, as they are both very important topics where the health of both mother and child can rely on thoroughly understanding this vital information!
This article aims to plainly spell out what MTHFR actually is in the scope of preconception and pregnancy, both highlighting its general role in the body, and a vital role both before and during pregnancy.
So, What Is MTHFR
MTHFR stands for methylene-tetrahydrofolate reductase.
It is an enzyme in your body that converts the folate you eat (like in leafy greens and legumes) into the active form called 5-MTHF, or, 5-Methyltetrahydrofolate.
This means, that if you have a mutation in your MTHFR gene, the ability for your body to create healthy levels of active folate will be affected or decreased. This will result in less active folate available for your body to use in several very important processes.
This is where the link between MTHFR gene mutation, preconception and pregnancy arises. Several of the vital processes that we need active folate for are highly active during times of preconception and pregnancy and need to be working well to support a healthy pregnancy.
During preconception and pregnancy, you need good levels of active folate for:
- Creating healthy DNA for both you and your future child. This is the biggest reason why knowing your MTHFR gene result is so important. Active folate is directly involved in the synthesis of new DNA. And while we have a constant demand for the production of new and healthy DNA, you can imagine that demand for this hugely increases during pregnancy, when you are growing a new life! Issues with not enough healthy DNA available for both mother and growing child can result in issues with pregnancy, fetal growth, and general childhood development.
- Preventing levels of a substance in the body called homocysteine from climbing too high, which can be related to blood clots and increased risk of blot clot formation during pregnancy.
- To create molecules called ‘methyl groups’, which act as instruction manuals for your DNA and cells, telling them the correct way to ‘behave’, so they do not do anything unwanted (e.g. cause disease or dysfunction within the body). We need healthy levels of these methyl groups to methylate/instruct your DNA, and without it cells are uncontrolled and can start to cause problems.
- Formation of red blood cells, white blood cells, and platelets, which are all vital for both the health of the mother during pregnancy and also for the health of the child during pregnancy and after birth as they begin to rapidly grow and come into contact with bacteria and pathogens to strengthen their immune system.
As you can see, addressing and supporting your MTHFR genes during your preconception phase is the best way to healthily support both your body once you fall pregnant, and the growth and development of your new baby.
Knowing your MTHFR gene result and supporting your folate levels where needed is a key step in preconception, and both should not be undervalued!
Carolyn Ledowsky,
Founder of MTHFR Fertility
PS: If you are looking for a more in-depth explanation and management of MTHFR before pregnancy, be sure to check out our Flagship course “MTHFR and Preconception“. This 4 to 8 module course will guide you through the ins and outs of preparation your body will need to have a healthy and thriving pregnancy. Modules include what tests to expect, how to analyze the results and steps to take to prepare mentally, physically and emotionally for your pregnancy.”
How Does Folic Acid Supplementing or Fortifying Affect Your Body?
There is a lot of buzz around folates and folic acid.
Health professionals often find the terminology around folate and folic acid confusing. They use the words folic acid supplements when they mean folate, folinic acid or 5-methyltetrahydrofolate (active form). The mix up occurs because folate has become an umbrella term for many forms of folate. Each study may use a specific form of folate such as 5-methyltetrahydrofolate but by the time the study makes it to the media, for convenience, it’s being referred to as folate. This creates a game of broken telephone lines between the researcher and the public, leading to confusion.
There are many forms of folate. Fortunately, you only need to know about the forms of folate relevant to you. It is easy to get confused, so we are going to walk you through why each form of folate is important.
Is Folic Acid Equal To Folate?
No!
Folates are found naturally in food and folic acid is a man made synthetic form. The body prefers to use naturally occurring folates over the folic acid. Folic acid is a stable synthetic compound making it easy to fortify food with. Folic acid can be found in supplements and fortified foods (ex. Bread, baked goods, cereals or any other foods made with folate enriched wheat). We need less than 200 micrograms of folate per day! But, if you have an MTHFR mutation your body will often require more than 200 micrograms of folate per day.
Folic acid has no function in your body until it is made into dihydrofolate. Having too much folic acid in the system blocks the pathway for naturally occurring folates. Folic acid inhibits the DHFR enzyme from making natural folates into dihydrofolate.This is why folic acid supplements are usually a bad idea. Not until recently, due to advances in technology, 5-methyltetrahydrofolate and folinic acid became available for supplementation.
Supplementing with 5-methyltetrahydrofolate or folinic acid is better than folic acid. Depending on your unique situation a folate supplement may not be the best option for you or may require different supplementation strategies. A combination of 5-methyltetrahydrofolate and folinic acid at ratios specific to your condition may be recommended or some of you will do better on just active methyl folate. Working with a healthcare practitioner who is specialized in working with clients with MTHFR mutations is recommended because not getting the ratios right can lead to more health problems.
For more information on MTHFR and folic acid and folic acid supplements read our article “Why isn’t folic acid good for those with an MTHFR mutation? ” and for more information about supplementing folate read “A guide to folate supplementation”
https://www.mthfrsupport.com.au/folate-and-brain/
https://www.mthfrsupport.com.au/folic-acid-vs-5-mthf-treating-mthfr-deficiency/
https://www.mthfrsupport.com.au/top-20-folate-containing-foods/
https://www.mthfrsupport.com.au/folic-acid-vs-5-mthf-debate/
Why Is Folate Important?

Folate is widely studied because it plays many critical roles within the body. Folate plays a role in the following processes:
- Synthesis of DNA and RNA
- Use of S-adenosyl methionine (SAM) for the transfer of methyl groups
- Production and recycling of neurotransmitters
- Detoxification
- Creation of functional red blood cells (RBC), white blood cells (WBC) and blood platelets
A lack of folate can lead to harmful conditions, especially before or during pregnancy. Low folate levels are linked to:
- Neural tube defects
- Anencephaly
- Spina bifida
- Encephalocele
- Poor quality sperm
- Blood clotting issues leading to increased risk of thrombosis
- Autism
- Behavioral issues such as ADD/ADHD
- Increased sensitivity to allergens
- Low birth weight caused by
- Premature birth
- Slow prenatal growth rate
- Multiple miscarriages
Folinic acid (also known as calcium folinate or 5-formyltetrahydrofolate) is a cofactor in many reactions playing a role in:
- Purine synthesis
- Pyrimidine synthesis
- Amino acid conversion
- All are important for synthesis of DNA
5-methyltetrahydrofolate is the active form of folate in the body. Once made, it plays a role in the processes mentioned above but it also plays a vital role in the homocysteine cycle.
If 5-MTHF is out of balance you can start running into problems. Supplementing 5-methyltetrahydrofolate without professional guidance may lead to biochemical imbalances within your body. These imbalances can cause a variety of side effects including:
- irritability
- insomnia
- sore muscles
- achy joints
- acne
- rash
- severe anxiety
- palpitations
- nausea
- headaches
- migraines
- depression
- suicidal thoughts
What Are Natural Sources Of Folate?
Folate primarily comes from some of the healthiest foods you can eat. Dark green leafy greens. Here is a list of foods known to be high in folate:
- Spinach
- Kale
- Swiss Chard
- Collard greens
- Romaine lettuce
- Brussel sprouts
- Asparagus
- Broccoli
- Cauliflower
- Avocados
- Okra
Folate can also be found in some nuts, lentils, and beans. For a more detailed article about folate containing foods including folate-fortified foods, click here.
Overview Of The Folate Cycle
The diagram below shows a simplified process of the necessary steps to turn folate to its active form 5-MTHF within the body.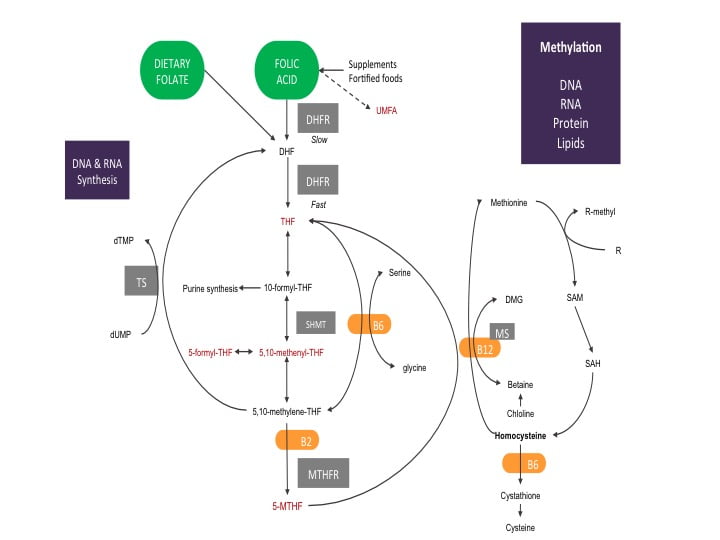
Starting with dietary folate, you can see it is ready to jump right into the folate cycle. The folate you eat is quickly made into dihydrofolate (DHF). DHF is given electrons by the enzyme dihydrofolate reductase (DHFR) using one molecule of nicotinamide adenine dinucleotide phosphate (NADPH, a common electron donor within the body and participates in many biochemical reactions) to make tetrahydrofolate (THF).
The diagram shows DHFR is quicker at making THF from dietary folate compared to folic acid. This is because the DHFR enzyme has more attraction to dietary folate and only needs to help with one reaction. Folic acid requires two reactions with the DHFR enzyme to become THF. The first reaction of folic acid is slow. The slow reaction process uses up available DHFR enzymes, blocking them from using natural folates.
The more folic acid present within the system, the slower folate metabolism becomes. If the system becomes saturated with folic acid, some research suggests DHFR enzymes can become completely blocked. A building up of folic acid begins, causing the folate cycle to become out of balance. Anytime an important biochemical process within the body becomes out of sync with its natural cycles there are negative health consequences. If you are trying to become pregnant or are experiencing treatment-resistant health issues, bringing the folate cycle back into its natural cycle is important for ensuring a healthy pregnancy and restoring the body’s methylation system. For a more in-depth article on the folate cycle read our article “The Folate Cycle In Detail”
Changing Your Relationship To Folate
There are major differences between types of folates. Keeping this in mind when choosing what to eat throughout the day. Avoiding foods fortified with folic acid is a good place to start. Unfortunately, folic acid fortification is mandatory in 53 countries around the world. Food such as bread, baked good, cereals are made with enriched wheat and are going to be fortified with folic acid. Making the choice to substitute folate fortified food for food containing natural folates can make a big difference in maintaining a healthy and functioning folate cycle.
Make sure you are including more dark leafy greens in your diet. Refer to our folate containing foods list to get a general sense of what contains folate when planning out your meals. MTHFR mutations decrease your ability to process folate. If you have an MTHFR mutation, avoiding foods fortified with folic acid and getting your natural folates into your diet should be a priority for you. If your health conditions persist after improving your dietary folate intake, you should speak with your healthcare practitioner about supplementing with a combination of active folates. Your situation may be more complex, requiring a specific supplementation strategy to restore a natural and healthy folate cycle.
You can join Carolyn for her next FREE webinar on “What is MTHFR?” for more information about MTHFR and folate. Watch our Events page for updates!
Take home message
The key message here is that we should avoid folic acid where possible, particularly in supplement form and fortified foods, particularly if you have an MTHFR gene mutation. You should be getting as much folate as you can from your diet in the way of leafy green vegetables, and if you have health conditions speak to your practitioner about supplementing with various combinations of active folates ie: folinic acid or 5-MTHF.
References
[1] Aarabi, M., San Gabriel, M. C., Chan, D., Behan, N. A., Caron, M., Pastinen, T., … & Trasler, J. (2015). High-dose folic acid supplementation alters the human sperm methylome and is influenced by the MTHFR C677T polymorphism. Human molecular genetics, 24(22), 6301-6313.
[2]Hekmatdoost, A., Vahid, F., Yari, Z., Sadeghi, M., Eini-Zinab, H., Lakpour, N., & Arefi, S. (2015). Methyltetrahydrofolate vs Folic Acid Supplementation in Idiopathic Recurrent Miscarriage with Respect to Methylenetetrahydrofolate Reductase C677T and A1298C Polymorphisms: A Randomized Controlled Trial. PloS one, 10(12), e0143569.
[3] https://www.cdc.gov/ncbddd/folicacid/recommendations.html
[4] https://ods.od.nih.gov/factsheets/Folate-HealthProfessional/
[5] Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. (1998). Dietary reference intakes for thiamin, riboflavin, niacin, vitamin B6, folate, vitamin B12, pantothenic acid, biotin, and choline. National Academies Press (US).
[6] Jacques, P. F., Bostom, A. G., Williams, R. R., Ellison, R. C., Eckfeldt, J. H., Rosenberg, I. H., … & Rozen, R. (1996). Relation between folate status, a common mutation in methylenetetrahydrofolate reductase, and plasma homocysteine concentrations. Circulation, 93(1), 7-9.
A Century-Old Procedure Offers New Hope
Does Flushing The Fallopian Tubes Increase Fertility?
Recently, infertility was brought into the spotlight throughout the medical community. Specifically, they were highlighting a century’s old procedure and the success rate experienced by couples trying to conceive around the world: flushing the fallopian tubes.
This is an old procedure that is over 100 years old. The technical name for this procedure is “Hysterosalpingography (HSG). The procedure involves flushing the fallopian tubes with iodized poppy seed oil and then undergoing an X-ray to get images of the uterus and fallopian tubes.
Sometimes the medical community gets lucky and stumbles upon new scientific discoveries, HSG is a great example of this. It was originally intended to be an imaging technique, but it turns out HSG might be making women more fertile!
In a study done in Canada, 1,119 infertile women underwent HSG treatment and 40% of the women who received HSG treatment with poppyseed oil fell pregnant within 6 months compared to 29% of women who received an HSG with water. That is a big difference in pregnancy rates between HSG poppyseed oil and HSG water treatment groups. The poppyseed oil group experienced a conception rate that was 11% higher than the water HSG treatment group.
Many of our patients at MTHFR Support and Fertility are using this procedure in combination with our preconception program with great success. You can read more about this procedure and the research at Medical News Today.
Low Homocysteine Levels: What are The Consequences?
What Is Homocysteine?
Homocysteine is an amino acid derivative that serves as an intermediate in the synthesis of methionine and cysteine. Low homocysteine develops when methionine, cysteine and folate levels become disrupted. Homocysteine contains a sulfhydryl group that serves as an important branch in the formation of important biological compounds such as glutathione (GSH) and S-adenosylmethionine (SAM). GSH is formed through the transsulfuration (TS) pathway with the formation of cysteine from homocysteine by a stepwise process by the enzymes cystathionine-beta-synthase (CBS) and cystathionine-gamma-lyase (CGL), and by subsequent formation of GSH by glutathione synthetase. SAM is formed by methylation of homocysteine to form methionine by the enzyme N5,N10-methylenetetrahydrofolate reductase (MTHFR) and subsequent condensation of methionine with adenosine triphosphate (ATP) to form SAM.
What You Should Know About Low Homocyesteine
Low homocysteine levels, or hypohomocysteinemia, is the result of several factors. It can be a result of a metabolic insult which prompts the body to produce more GSH than normal, low intake of the amino acids methionine and cysteine (methionine predominantly comes from meat protein so those that don’t eat meat or eat too little may be at higher risk), an inherently low level of the enzyme MTHFR, low intake of vitamins folate and B12, or increased detoxification of xenobiotics through the phase II liver reaction sulfation. These factors will result in low homocysteine levels, which would then lead to a variety of clinical disorders.
Hypohomocysteinemia has been shown to play a role in the malnutrition-inflammation-cachexia syndrome with which low dietary intake of methionine would lead to worsening of chronic kidney disease, with which homocysteine can be a variable in determining survivability of the patient. Hypohomocysteinemia is also a cause of the progression of atherosclerosis in cardiovascular diseases.
Since hypohomocysteinemia may also present with a lack of cysteine levels, it can be due to the role that the liver plays. Synthesis of GSH requires cysteine, in which the formation of GSH will get homocysteine from the reservoir to support the body in times of oxidative stress, in which GSH may be able to mediate and prevent oxidative damage to cells. The synthesis of GSH would, therefore, favor the TS pathway. So basically this means if there is a low level of antioxidants, the body will steal cysteine from homocysteine to support glutathione production. So low homocysteine may be a sign of oxidative stress.Formation of more cysteine from homocysteine would then deplete the homocysteine pool to support the formation of GSH to combat the free radicals.
Another factor that would deplete homocysteine levels would be the detoxification pathway sulfation. Sulfation is a phase II or conjugation reaction in which cysteine donates a sulfur group to the xenobiotic forming a xenobiotic-sulfate, allowing it to be excreted from the system. Apart from sulfation, cysteine is also employed in the formation of bile acids such as taurine. Taurine is especially synthesized when the body takes in fat and alcohol. Synthesis of taurine would also deplete homocysteine levels by promoting the TS pathway in the formation of cysteine. These homocysteine-depleting mechanisms are prevented if there are adequate amounts of methionine and cysteine from the diet.
Low dietary intake of vitamins folate and B12 can also lead to hypohomocysteinemia. Folate is an important component of the methylation pathway as it forms the structure of N5,N10-methylenetetrahydrofolate and would, therefore, be an important substrate for the transmethylation pathway involving the formation of SAM by transferring a methyl group from N5,N10-methylenetetrahydrofolate to homocysteine forming methionine, catalyzed by the enzyme MTHFR. Folate is important in regulating gene expression by regulating levels of SAM.3
Vitamin B12 is another vitamin that is important in gene expression and DNA synthesis. Methylcobalamin which serves as a cofactor in the methylation cycle is involved in the production of methionine from homocysteine by the action of MTHFR. Low dietary intake of either or both folate and vitamin B12 will result in low methionine levels, which would ultimately inhibit the formation of homocysteine.4
MTHFR deficiency may also lead to low homocysteine levels in the body. MTHFR is the enzyme responsible for the transfer of a methyl group from N5,N10-methylenetetrahydrofolate to homocysteine forming methionine. Defects in the gene, most notably the C677T gene polymorphism, may result in low 5-MTHF levels, resulting in hypohomocysteinemia.5
The effects of having hypohomocysteinemia are mainly due to the prolonged depletion of cysteine. That said, the effects of hypohomocysteinemia would be the prolonged inflammatory status experienced by the body due to the increased formation of free radicals with the lack of GSH to counteract the effect. Inflammation can lead to several disorders that may affect the cardiovascular system, nervous system, and even the renal system. Atherosclerosis is a major concern due to the inflammatory nature of the disease. Inflammation will induce the misfolding of the proteins in the brain causing Alzheimer’s disease. Chronic renal failure may result in the increased inflammation of the tubular system. GSH plays a central role in mediating the effects of oxidative stress, preventing damage to the organs. That said, GSH production relies heavily on the pool of homocysteine to fuel its synthesis.
REFERENCES:
- Ganguly, P. & Alam, S.F. Role of homocysteine in the development of cardiovascular disease. USA: Nutrition Journal. January 2015; Vol. 14, No. 6.
- Lord, R. & Fitzgerald, K. Significance of Low Plasma Homocysteine. USA: Metametrix Laboratory Department of Science and Education. 2006. Retrieved https://www.drkendalstewart.com/wp-content/uploads/2011/09/Significance-of-Low-Plasma-Homocysteine.pdf
- Blom, H. & Smulders, Y. Overview of homocysteine and folate metabolism. With special references to cardiovascular disease and neural tube defects. USA: Journal of Inherited Metabolic Disorders. February 2011; Vol. 34, No. 1. pp. 75-81.
- O’Leary, F. & Samman, S. Vitamin B12 in Health and Disease. USA: Nutrients. March 2010; Vol. 2, No. 3. pp. 299-316.
- Leclerc, D. et al. Molecular Biology of Methylenetetrahydrofolate Reductase (MTHFR) and Overview of Mutations/Polymorphisms. Madame Curie Bioscience Database.
Taking Folic Acid When You Have An MTHFR Mutation | Why Is It Not Recommended
So you have one of the common MTHFR mutations and are not sure how to start supplementing. You went online to find out what to take and read that taking folic acid is a good idea. We are here to tell you it’s NOT. Check out our article “Using Folic Acid Supplements In Fertility Treatments” for an overview. Once you are up to speed on folic acid, take a look at where it enters the folate cycle in the graphic below.
You can see folic acid entering the folate cycle right at the beginning. It’s the first molecule setting the folate cycle in motion, but it’s not the best molecule to start the process of folate metabolism. This type requires an extra metabolic step, that is slow and inefficient, before entering the folate cycle where naturally occurring folates do.
Folic Acid Vs. Natural Folates
Folic acid is a synthetic form of folate (vitamin B9) that are made to be more chemically stable. It is made to be more stable than naturally occurring folates because they are not broken down during the manufacturing process as much. Companies can get folic acid to the shelf a lot easier than they can get naturally occurring folates. The companies want to make as much money as possible so they market and sell synthetic product over naturally occurring folates. Extracting, processing and storing naturally occurring folates is difficult and expensive. Many of the folates at the beginning of the manufacturing process end up breaking down, rendering them useless. Folic acid was the first form of folate available but in recent years the active forms are now able to be put in supplements.
Dihydrofolate Reductase (DHFR)
The dihydrofolate reductase (DHFR) enzyme breaks down folic acid and natural folates into tetrahydrofolate (THF). Taking folic acid can reduce your ability to metabolize naturally occurring folates you are getting through your diet. The graphic shows DHFR acting slowly on folic acid and fast on dietary folates. This is because it is made to be a more stable molecule compared to non-synthetic forms of folate. The chemical stability and slight change in shape of folic acid compared to natural folates, make it difficult for DHFR to use/metabolize. The reaction takes much longer to happen because the chemical structure of folic acid is resistant to metabolism by DHFR.
There are a limited number of DHFR enzymes available for metabolizing folic acid and dietary folates. When most of the folates in your diet are coming from synthetic supplementation or from foods fortified with folic acid, the DHFR enzymes are busy trying to metabolize folic acid instead of naturally occurring folates.
The DHFR enzyme itself can only metabolize 200 mcg of folic acid per day. If you are getting over 200 mcg of folic acid through supplementing and/or from fortified foods, the DHFR enzyme is going to be inhibited. This causes a build of folic acid and folates at the beginning of the folate cycle within the body. The build-up of folic acid is called un-metabolized serum folic acid or UMFA for short.
For more information about UMFA read our article Folic Acid: What Happens When You Have Too Much?
Take Aways and MTHFR
Too much folic acid in your diet from fortified foods, or taking folic acid supplements, is going to reduce your ability to metabolize folates you are getting from your diet. It is best to stay away from foods fortified with folic acid when you can and eat more food with high levels of naturally occurring folates. Since folic acid will build up in the folate cycle, there will be less folate making it through the final stage of the cycle which requires the MTHFR enzyme. If you have an MTHFR mutation, you know your MTHFR enzyme has a 20-70% loss in function when converting 5,10-methylene-THF to 5-MTHF. Including folic acid in your diet or supplement makes the entire folate cycle even less efficient! You need to avoid taking folic acid and adopt a diet full of natural folates, especially if you have an MTHFR mutation! Here you can find out the Top 20 Folate Containing Foods.
For some people, a consistent and high folate diet may be difficult. If you are worried about not getting enough folates, there are better ways to get folates through supplementation than through a synthetic method, check out our Guide To Folate Supplementation.
If you are preparing your body for a healthy and thriving pregnancy, we have a free 10-day email course we recommend to get you started on the right path. Sign up for free today and get started on the best path for you and a thriving pregnancy.
High homocysteine – it can affect your brain, your heart and your pregnancy.
High homocysteine – it can affect your brain, your heart and your pregnancy.
Hyperhomocysteinemia, the condition of having high homocysteine in the blood, causes numerous health issues within the body. This article will cover health and pregnancy-related aspects of high homocysteine. If you do not know what homocysteine is or want to know more about homocysteine before reading this article check out “What’s All The Fuss About Homocysteine?”
High homocysteine levels cause hyperhomocysteinemia — a condition that results in fertility issues, mental health disorders, and cardiovascular problems. Since individual genes are responsible for metabolizing homocysteine, people with MTHFR mutations and mutations in genes involved in vitamin B and folate metabolism are at risk of developing high homocysteine levels.
High homocysteine can be treated with a combination of nutrients like folate, B12, B6 and trimethylglycine. It’s important to work with a practitioner because levels can drop quickly and the formula may need to be changed frequently to avoid homocysteine levels dropping too low.
Homocysteine and Cardiovascular Disease
High homocysteine concentrations increase one’s risk for cardiovascular disease. Arteries harden and narrow due to hyperhomocysteinemia, ultimately resulting in cardiovascular disease – which occur in coronary, cerebral and peripheral blood vessels. People with a familial history of, or current, cardiovascular disease should get tested frequently and seek medical advice.
Homocysteine and Mental Illness
According to studies, the nervous system can be severely harmed by high levels of homocysteine in the body. Homocysteine functions to maintain blood vessels, is a vital intermediate molecule in the creation of primary methyl donors. High levels of homocysteine results in numerous biochemical imbalances in the body, and in the brain, which causes irreparable damage to our bodies and mental health.
A study has shown that patients with depression also had hyperhomocysteinemia, metabolic disorders involving folate metabolism, and neurotransmitter metabolic issues. It proved that 20-30% of patients suffering from hyperhomocysteinemia and depression showed more severe symptoms. It is therefore evident that hyperhomocysteinemia does not affect everyone in the same manner. A specific supplementation program is essential to determine whether your hyperhomocysteinemia is affected by depression. However, depression is not the only condition related to mental health that could show exacerbated symptoms due to high levels of homocysteine. Homocysteine levels are known to increase as we age. Dementia and Alzheimer’s are also potential resultant effects from such increased cellular concentrations.
Homocysteine and Pregnancy
Homocysteine levels during pregnancy are relatively inconsistent as they continuously fluctuate. However, high amounts of homocysteine during pregnancy can result in adverse outcomes to both mother and child. Additionally, some people may experience changes due to insufficient vitamin cofactors or have impaired functionality of methionine synthase.
Methionine is an enzyme which converts homocysteine to methionine. This process is called transsulfuration or remethylation. When transsulfuration is not working or inactive, it can lead to myelin deficiency during pregnancy (Mills, 1995). Myelin is a fatty material, white in appearance which forms a sheath around the neurons within the central nervous system. The purpose of myelin is to increase the speed of neural transmission and is crucial for the proper function of the nervous system. Multiple Sclerosis (MS), is an autoimmune disease that attacks and destroys the myelin surrounding neurons. This damage can lead to a wide variety of symptoms associated with MS that include:
- cognitive impairment
- depression
- dizziness
- optic neuritis
- pain
- fatigue
- tinnitus
- muscle weakness
- numbness/tingling
- tremors
- twitching
Neural tube defects or NTD’s, which are birth defects of the brain, spine or spinal cord, can arise when there is not enough myelin production in a fast-growing fetus during pregnancy. The most significant development of myelination occurs during the third trimester of fetal development and progresses rapidly during the first few years of a child’s life. Environmental input supportive of myelination during pregnancy does not only help prevent neural tube defects but also helps to develop a functional and healthy nervous system for your child to inherit.
Pre-eclampsia
When there is too much homocysteine in the blood it can cause damage to the vascular system, which in turn may lead to increases in blood pressure, vascular abnormalities in the mother and fetus, and ultimately preeclampsia. Although the cause of preeclampsia remains largely unknown., it appears to be caused by oxidative stress during pregnancy from high levels of homocysteine.
Research surrounding preeclampsia and homocysteine shows evidence supporting the harmful role hyperhomocysteinemia plays in the development of preeclampsia. A study done by Dekker et al., 1995, found that 2% of pregnant women have pregnancies that develop pre-eclampsia, and 17.7% of these women have hyperhomocysteinemia. A follow-up study done by Leeda et al., in 1998, confirmed the finding of Dekker and his team – showing the association between preeclampsia and hyperhomocysteinemia.
Placental Abruption
Placental abruption (Abruptio placentae) occurs when the placental lining detaches from the uterus (after week 20 and before birth), causing internal bleeding. Depending on the severity, the mother and the fetus could face death or serious injury. Placental abruptions occur, on average, 1 in 200 pregnancies and are a major contributor to maternal deaths around the world.
In a study done by Goddijin-Wessel et al., in 1996 found 31% of pregnant patients who have placental abruptions have hyperhomocysteinemia after a methionine loading test, compared to 9% of the control group. Methionine loading tests provide insight into how efficiently the body is utilizing homocysteine. This study concluded that higher levels of homocysteine result in higher chances of having placental abruptions.
Another study was done by Vries et al., in 1998 found that 26% of people with placental abruptions without preeclampsia had hyperhomocysteinemia 3 months after their pregnancy. This study concluded that having a placental abruption or preeclampsia during pregnancy can be an indicator of hyperhomocysteinemia. In such a case, you should get your homocysteine levels checked if you experience these complications during pregnancy.
Thromboembolic Events
Hyperhomocysteinemia is a subcategory of a broader set of congenital conditions called thrombophilias. Thrombophilias occur when the body has an abnormal tendency of blood to clot. This results in higher chances of thromboembolic events occurring, namely, heart attacks, strokes and pulmonary embolisms. Unfortunately, hyperhomocysteinemia is more likely to cause thromboembolic events in women compared to men. Women are 3.8 times more likely to have a thromboembolic event compared to men having a 1.8 times higher chance.
Pregnancy itself is a risk factor for thromboembolic events, further increasing the risk in addition to hyperhomocysteinemia. With the right precautions, the risk of having a thromboembolic event during pregnancy can be mitigated. Pregnancy and hyperhomocysteinemia are not the only risk factors for thromboembolic events. All factors should be taken into consideration while developing a plan to decrease or prevent chances of thromboembolic events before, during and after pregnancy.
Fetal Death in Utero
The conditions leading to fetal death are complex and come from many sources. Often a combination of embryonic processes go wrong, many of which are outlined in this article. Preventing fetal death requires a multifaceted approach which can be overwhelming if taken on alone. If you are worried, had previous issues, or are at risk, then reach out for professional support who will conduct proper examinations through regular check-ups and give advice during every step of your pregnancy.
Intrauterine Growth Retardation (IUGR)
Only a few studies have been done around the world into IUGR and hyperhomocysteinemia. The results of these studies obtained are somewhat contradictory. Researchers hypothesize that hyperhomocysteinemia increases the likelihood of IUGR and their studies both support and reject their hypothesis. More research is needed to clear up the contradictory scientific evidence gathered so far. It is too soon to jump to any conclusions about the relationship between hyperhomocysteinemia and IUGR and larger prospective studies are needed in this field.
Recurrent Pregnancy Loss
The main hypothesis surrounding homocysteine and recurrent pregnancy loss is that high levels of homocysteine interfere with the implementation of the embryo into the endometrial wall of the uterus. This interference occurs due to premature damage to the capillaries that supply the endometrial uterine wall. The capillaries are less functional due to excessive amounts of homocysteine being present, making them unable to support the new embryo and leading to miscarriages. In animal models, using rats and chickens, homocysteine levels higher than 15 ɥmol/l cause 75% of pregnancies to abort due to developmental abnormalities.
There are significant links between recurrent pregnancy loss and hyperhomocysteinemia, 14% of women who have primary repeated miscarriages have hyperhomocysteinemia and 33% of women who have secondary repeated miscarriages have hyperhomocysteinemia. If you have had miscarriages in the past, or are worried about having miscarriages in the future, make sure to get your cellular homocysteine concentrations to the correct levels.
Take Aways
There is overwhelming scientific evidence suggesting high homocysteine (hyperhomocysteinemia) increases fertility complications, cardiovascular disease and mental health issues. The most pressing and immediate concerns of hyperhomocysteinemia are its effects on pregnancy. If you are considering pregnancy, make sure you check homocysteine levels before, during and after pregancy, especially if you have the MTHFR gene mutation as this increases the risk of having high homocysteine levels. Creating a strategy with a trained professional is an even better idea because pregnancy is an extremely complicated process with a lot of moving parts. Ensuring you have professional support during pregnancy is in you and your child’s best interest. High homocysteine levels are not the only problem, you could be experiencing health problems from low homocysteine levels.
References
(1) Aubard, Y., Darodes, N., & Cantaloube, M. (2000). Hyperhomocysteinemia and pregnancy—review of our present understanding and therapeutic implications. European Journal of Obstetrics & Gynecology and Reproductive Biology, 93(2), 157-165.
(2) Dekker, G. A., De Vries, J. I. P., Doelitzsch, P. M., Huijgens, P. C., Von Blomberg, B. M. E., Jakobs, C., & Van Geijn, H. P. (1995). Underlying disorders associated with severe early-onset preeclampsia. American journal of obstetrics and gynecology, 173(4), 1042-1048.
(3) Den Heijer, M., Koster, T., Blom, H. J., Bos, G. M., Briët, E., Reitsma, P. H., … & Rosendaal, F. R. (1996). Hyperhomocysteinemia as a risk factor for deep-vein thrombosis. New England Journal of Medicine, 334(12), 759-762.
(4) Goddijn-Wessel, T. A., Wouters, M. G., vd Molen, E. F., Spuijbroek, M. D., Steegers-Theunissen, R. P., Blom, H. J., … & Eskes, T. K. (1996). Hyperhomocysteinemia: a risk factor for placental abruption or infarction. European Journal of Obstetrics & Gynecology and Reproductive Biology, 66(1), 23-29.
(5) Hague, W. M. (2003). Homocysteine and pregnancy. Best practice & research Clinical obstetrics & gynaecology, 17(3), 459-469.
(6) Leeda, M., Riyazi, N., de Vries, J. I., Jakobs, C., van Geijn, H. P., & Dekker, G. A. (1998). Effects of folic acid and vitamin B 6 supplementation on women with hyperhomocysteinemia and a history of preeclampsia or fetal growth restriction. American journal of obstetrics and gynecology, 179(1), 135-139.
(7) Mills, J. L., Lee, Y. J., Conley, M. R., Kirke, P. N., McPartlin, J. M., Weir, D. G., & Scott, J. M. (1995). Homocysteine metabolism in pregnancies complicated by neural-tube defects. The Lancet, 345(8943), 149-151.
(8) Refsum, MD, H., Ueland, MD, P. M., Nygård, MD, O., & Vollset, MD, Dr. PH, S. E. (1998). Homocysteine and cardiovascular disease. Annual review of medicine, 49(1), 31-62.
(9) Reynolds, E. H. (2002). Folic acid, ageing, depression, and dementia. British Medical Journal, 324(7352), 1512.
(10)Rosenquist, T. H., Ratashak, S. A., & Selhub, J. (1996). Homocysteine induces congenital defects of the heart and neural tube: effect of folic acid. Proceedings of the National Academy of Sciences, 93(26), 15227-15232.
(11)Vanaerts, L. A., Blom, H. J., Deabreu, R. A., Trijbels, F. J., Eskes, T. K., Peereboom‐Stegeman, J. H., & Noordhoek, J. (1994). Prevention of neural tube defects by and toxicity of L‐homocysteine in cultured postimplantation rat embryos. Teratology, 50(5), 348-360.
(12)Vries, J. D., Dekker, G. A., Huijgensb, P. C., Jakobs, C., Blomberg, B. M. E., & Geijn, H. V. (1997). Hyperhomocysteinaemia and protein S deficiency in complicated pregnancies. BJOG: An International Journal of Obstetrics & Gynaecology, 104(11), 1248-1254.
(13)Wouters, M. G., Boers, G. H., Blom, H. J., Trijbels, F. J., Thomas, C. M., Borm, G. F., … & Eskes, T. K. (1993). Hyperhomocysteinemia: a risk factor in women with unexplained recurrent early pregnancy loss. Fertility and sterility, 60(5), 820-825.
Polyphenols and Phenols – Prenatal Exposure – What you need to know & 5 Practical Tips
If you’re reading this, you probably already have some sense of what phenols are, and what they do. But if you have no idea what phenols are, here is a quick overview of Polyphenols and Phenols and Prenatal Exposure
What Are Phenols and Polyphenols?
Phenols are a large class of chemical compounds. They play a role in regulating cell growth and adipogenesis (The process of cells specializing to become fat cells), by interacting with nuclear receptor sites (the location where hormones collaborate with cells).
The hydroxyl group on phenols is what makes them have unique properties. They can bond tightly with oxygen and have a reactive oxygen-hydrogen bond at the same time. These chemical characteristics give rise to some potentially beneficial effects (from NATURALLY occurring phenols) within the body such as:
- Antioxidant
- Anti-ageing
- Cancer prevention
- Improved cardiovascular health
Types Of Phenols
There are two types of phenols you can find: natural and synthetic. The benefits come from naturally occurring phenols, which are absorbed into the body through the food you eat. Healthy, naturally occurring phenols come from fruits and vegetables, primarily in the form of flavonoids (a polyphenol). It is important to understand the difference between Polyphenols and Phenols and Prenatal Exposure. Polyphenols are the most beneficial phenols to consume. You can find high polyphenol concentrations in the following foods listed here.
Naturally occurring phenols and polyphenols are powerful compounds that are beneficial to the body but are not fully understood by science. You will never get too many polyphenols from over eating fruits and vegetables, so don’t worry about these phenols; worry about synthetic phenols!
Synthetic Phenols: What You Need To Know
What Do They Do?
Synthetic phenols may pose some health risks during pregnancy and to overall health. They have the ability to act as endocrine (hormonal) disruptors by interacting with the following hormone receptors [2]:
Glucocorticoid receptors
- Play a role in: development, metabolism and immune response
Thyroid receptors
- Play a role in: development, heart rate and metabolism
Estrogen receptors
- Play a role in: development, gestation, gene regulation, metabolism and behavior
Where Are They Found?
- Solar filters
- Cosmetics
- Antibacterial soaps
- Polycarbonate plastics
- Epoxy resins
Polyphenols and Phenols – Prenatal Exposure To Synthetic Phenols
Synthetic phenols may pose a risk to pregnancy. The risk of synthetic phenols to development and pregnancy arises from their ability to mimic important hormones within the human body. They interact with cell receptors, causing changes to basic processes within the body.
In 2012 and 2014, researchers looked at what effects prenatal exposure to synthetic phenols had on the growth of boys, during and after pregnancy [4,5]. They looked at: Benzophenone-3, Parabens, Triclosan, Bisphenol A and dichlorophenol levels at 12, 22.5 and 32.6 gestational weeks through urine sampling concentrations. The researchers analyzed how phenol concentrations were affecting biparietal diameter (BPD) during pregnancy. BPD is the standard measurement to assess fetal size. BPD measures: head circumference, abdominal circumference and femur length. They sampled 520 mother-son pairs from April 2003 to March 2006 at Poitiers and Nancy University hospitals in France. The results of their studies are summarized below:
Benzophenone-3
- Common ingredient in sunscreens and lotions
- No association with BPD during pregnancy but a few days after birth there is a positive association with head circumference
Parabens
- Common additive in many products (see link for more information)
- Higher paraben concentrations lead to increased weight at birth
- Methylparaben is positively associated with weight and abdominal circumference at 36 months
Triclosan
- Found in soaps, shampoos, deodorants, toothpastes, cleaning supplies and pesticides
- Reduces all measurements of BPD in late pregnancy
- Reduces head circumference at birth
- Head circumference is used to predict brain volume [1]
- In animal studies triclosan disturbs thyroid receptors which alters fetal growth and brain development [3]
- Found in plastics and epoxy resins
- Found no clear association between Bisphenol A and BPD growth parameters at birth or 6-months
- Positively associated with weight at 12, 24 and 36 months
- Positive association with waist circumference, BMI and risk of being overweight at the age of 4 but not earlier [6]
Dichlorophenol
- Found as an intermediate chemical in advanced chemical manufacturing processes for the production of herbicides
- Negatively associated with abdominal circumference at 32.6 gestation weeks and positively associated at 36 months
- Positive association with weight between 24 and 36 months but not after 36 months
***Negative association means that as the concentration of the phenol increases, the growth parameter decreases***
***Positive association means that as the concentration of the phenol increases, the growth parameter increases***
5 Practical Steps To Reducing Exposure To Synthetic Phenols
- Write a list of synthetic phenols and check the ingredients of products around your house. When it comes time to replace the product, try and one that does not contain a synthetic phenol on your list.
- Use water bottles and Tupperware that are BPA free
- Buy fruit and vegetables that have no or low exposure to herbicides
- Use natural, synthetic phenols free cosmetic products when possible
- Shampoo
- Deodorant
- Make-up
- Lotions
- Buy Triclosan free cleaning products
Take Aways
The relationship between phenols and pregnancy is in its infancy. Some studies contradict each other, but general trends are emerging about how different phenols can have an effect on biparietal diameter (BPD). Properly measuring phenols are difficult because phenol measurements are made based on urine samples, and certain phenols will degrade before they can be accurately measured (short half-life), plus phenol release from the body is periodic. Due to the volatility and unpredictability of phenols in urine samples, studies looking at how phenol exposure relates to pregnancy are likely subject to exposure misclassification, meaning that researchers may be drawing the wrong conclusions from their data. We need bigger studies with larger samples sizes and better techniques for measuring phenol concentrations.
Even if we are still waiting on more research to happen before drawing final conclusions surrounding phenols and pregnancy, is it worth the risk to take no action towards reducing phenol exposure if you are looking to become pregnant or are currently pregnant? The answer is no! Phenols play a complex role in the body by mimicking important hormones. At high enough concentrations phenols pose, not only a risk to your pregnancy, but to your general health!
If you are looking for a more in-depth explanation and management of MTHFR before pregnancy, be sure to check out our Flagship course “MTHFR and Preconception”.
This course includes information on compounds to avoid, a comprehensive look at the prenatal process and will guide you through the ins and outs of preparation. Modules include what tests to expect, how to analyze the results and steps to take to prepare mentally, physically and emotionally for your pregnancy. For more information, check it out here.
Folate Supplementation Guide
Folate Supplementation Guide
You are looking to get pregnant and want to do all you can to ensure you have a happy and healthy baby. When done correctly, folate supplementation can make a huge impact on your child’s health during pregnancy!
The word folate is an umbrella term, describing multiple forms of folate. Using the term folate keeps things simple, but when you dig deeper beyond the umbrella you discover folate has many faces.
There are three forms of folates you should be aware of when beginning to think about folate supplementation.
- Folic Acid
- Folinic Acid
- 5-MTHF
This article will explain the basics of these 3 forms of folate for supplementation purposes. You will learn about which supplements help move the folate cycle along, what advantages each supplement has over others, what folate supplements to stay away from, and when to begin taking folate supplements.
When To Supplement with Folate
Folate supplementation should start long before pregnancy begins. Ensuring proper folate levels during preconception preparation is something everyone hoping to become pregnant should do. It takes time for the body to get the folate cycle back up and running efficiently (at least a couple weeks). If there is a problem such as a MTHFR mutation or other problems affecting the folate cycle, it may take longer to normalize. Folate supplementation should begin during preconception and continue throughout the entire pregnancy.
You want to prevent your body from having low folate levels long before you reach the periconceptional period of your pregnancy. During the first 3-4 weeks of pregnancy (periconceptional period), low folate levels play a role in the development of neural tube defects (NTD’s) and many other pregnancy complications, which are listed in our article “What is folic acid?”.
Folic Acid (Not Recommended)
The daily recommended intake of folic acid is 400 mcg. Common multivitamins will give you around 400 mcg of folic acid. If you are taking a multivitamin for folic acid, taking another folic acid supplement is going to be overkill. You will be getting too much folic acid. You and your child will be at risk for developing neurological disorders because folic acid at high levels is toxic to the human nervous system.
The one advantage folic acid has over other supplementation option is that it’s inexpensive. Folic acid is a synthetic compound that can be easily stored, transported and has a long shelf life. Other than being less expensive than other forms of folate supplementation, folic acid comes up short for the following three reasons:
- Folic acid is a synthetic compound that has NO physiological function until converted to dihydrofolate by dihydrofolate reductase (DHFR)
- The DHFR enzyme breaks down folic acid much slower than naturally occurring folates which causes a build up of folic acid.
- Folic acid has a stronger attraction to the folate receptors, blocking them from pulling natural folates into the cell for metabolic processes (our natural folates like leafy greens are essential for our folate levels)
When you take folic acid your body is not going to make use of the best folates available. This is due to the compounding of your cells folate receptors having a preference for folic acid and your enzymes being slower to react with folic acid. The DHFR enzyme has trouble metabolizing folic acid and on top of that, the DHFR enzyme is susceptible to mutations that reduce its functionality. If you have or think you have an MTHFR mutation, folic acid supplementation can create even more problems for your folate cycle. You can read more about “Why Folic Acid Is Not Good If You Have A MTHFR Mutation”
Folinic Acid (Recommended)
You can avoid the pitfalls of folic acid supplementation and/or a harmful DHFR mutation, by supplementing with folinic acid. Folinic acid enters the folate cycle in a better position than folic acid. Folinic acid or 5-formyltetrahydrofolate, enters directly into the middle of the folate cycle. It can be made into any of the possible products of the folate pathway, making it ideal for people who have DHFR mutations, or people who are exposing themselves to high levels of folic acid by consuming folic acid fortified foods.
Folinic acid directly contributes to physiological processes, unlike folic acid which has no direct physiological function. Folinic acid aids the synthesis of DNA by acting as a cofactor in the metabolic reactions responsible for creating purine and pyrimidine (two of the four building blocks of DNA).
There are three main points regarding folinic acid you should take away with you:
- Folinic acid can be made into everything folic acid or natural folates can be made into
- Folinic acid has direct physiological functions that help create the building blocks of DNA
- Folinic acid bypasses the DHFR enzyme in the folate cycle, making it an ideal supplementation strategy if you have a DHFR mutation that reduces your ability to metabolize folates and folic acid
5-MTHF or Active Folate (Recommended)
If you know you have an MTHFR mutation, 5-MTHF is going to be the most important supplement for you to take. 5-MTHF is the active form of folate that allows the body to recycle its methyl donors (the molecules responsible for turning off and on physiological processes within the human body by donating methyl groups).
The folate cycle is designed to produce 5-MTHF. 5-MTHF is the end product of the folate cycle and its production is directly affected by the MTHFR enzyme. Having an MTHFR gene mutation reduces your body’s natural ability to produce its active form of folate. When you read about the problems caused by a lack of folate, it’s due to a lack of the bodies active form of folate, 5-MTHF. Below is a quick recap of the problems linked to low folate status:
- Neural tube defects
- Lower sperm quality
- Blood clotting disorders (thrombophilia)
- Autism
- ADD/ADHD
- Allergies
- Low birth rate
An eye should be kept on your folate status if you are trying to become pregnant. During pregnancy, there are high volumes of DNA being produced every day. The child is growing rapidly and has the highest risk of being negatively affected by low folate levels.
The body is a highly complex system, and when you change your diet, your environment, or start supplementing to improve your folate status, it takes a couple months for your body to adjust to healthy folate levels.
Take Aways
Supplementing folates properly, by taking the right supplements (Folinic acid and 5-MTHF) and avoiding getting too much folic acid, will ensure you are managing your MTHFR mutation in a safe way. Leaving your MTHFR mutation as is, without taking steps to reduce its negative impact, is not good for you or pregnancy. Taking the necessary steps to ensure proper methylation throughout the body by maintaining sufficient folate levels through supplementation, if needed, will greatly reduce the risk of developmental health defects for your child.
Introduction To MTHFR Mutations
If you have heard of MTHFR mutations before but didn’t know where to start. This article is for you! It will give you a foundational understanding of biology while introducing you to MTHFR and fertility.
What Are Genes?
Genes are the deoxyribonucleic acid (DNA) sequences responsible for making proteins and enzymes. Gene expression is the process of the body actually using the gene to produce a protein or enzyme. Many factors contribute to gene expression and gene expression changes throughout life. For example, gene expression is different before puberty and after puberty, or before menopause and after menopause. Gene expression also changes due to psychological, environmental and dietary factors.
What Are Genetic Mutations?
DNA is made up of four building blocks: adenine (A), thymine (T), guanine (G) and cytosine (C). Genetic mutations are changes within a sequence of DNA. For example, an “A” might change to a “C”, or a “T” might be removed/added to a DNA sequence. There are multiple forms of genetic mutations that are beyond the scope of this article. It is important to know that genetic mutations mostly have negative consequences on the performance of their products (i.e enzymes).
What Are Enzymes and Why Are They So Important?
Enzymes are proteins that help build, break down, and/or speed up biochemical reactions within the body. Some chemical reactions would take days, weeks, or months to happen, but enzymes make these reactions happen much faster. Without enzymes, life as we know it would not be possible. Chemical reactions would be too slow within the body, and we would not have the capacity to produce the necessary chemicals for survival.
What Is MTHFR?
MTHFR stands for Methylenetetrahydrofolate reductase. The MTHFR enzyme is made from the MTHFR gene. The MTHFR gene contains the genetic information required to make the MTHFR enzyme. It is the enzyme responsible for the final step in the folate cycle. When the MTHFR enzyme is not functioning properly, there is a shortage of important biochemical substances and a build-up of harmful substances within the body. MTHFR gene mutations impair the body’s ability to perform many crucial tasks.
How Common are MTHFR Mutations?
Some people, approximately 30-50% but some research shows possibly 70-75%, have genetic mutations in their MTHFR gene (3). As more people have their genomes sequenced around the world, we will get a better understanding of how prevalent MTHFR mutations are around the world. Different populations of people around the world have different frequencies of MTHFR mutations, for example, the highest concentration of MTHFR C667T mutations in the world are found in Italy (1). These mutations affect the ability of the MTHFR enzyme to complete the final step in the folate cycle. MTHFR mutations lead to a complex cascade of biochemical reactions that cause a wide variety of health and fertility issues.
Consequences Of The MTHFR Mutations On Health And Fertility
According to a new scientific review published in July 2017, in the Journal of Genes and Genomics, MTHFR mutations play a role in the following multifactorial diseases (2):
- Cardio-cerebrovascular
- Neurodegenerative disorders
- Autoimmune diseases
- Diabetes
- Neuropsychiatric disorders
- Renal diseases
- Cancer
- Birth defects
The health consequences of MTHFR are so far-reaching because the MTHFR enzyme is crucial to basic functions within our bodies. If you are wondering how one gene/enzyme can have such a wide variety of consequences, without getting too involved in the science, you need to gain a basic understanding of how MTHFR mutations actually create health and fertility problems.
Understanding MTHFR Mutations
There is not just one kind of MTHFR mutations there are several (MTHFR C667T and A129C are most common). Each mutation lowers the functionality of the MTHFR enzyme to varying degrees. Having an MTHFR mutation is like your body building a single lane bridge in the middle of a busy city when the bridge should be at least four lanes wide. It significantly reduces the number of cars (i.e biochemical reactions) that can use the bridge at one time and creates a traffic jam on both sides (i.e build up of biochemical products).
Take Aways
Hopefully, you now have a basic understanding of MTHFR. If you are looking for more information on MTHFR, there are multiple articles surrounding how MTHFR Mutations affects your overall health and fertility that can be found below:
- High homocysteine – It can affect your brain, your heart and your pregnancy.
- Why is Knowing You Have the MTHFR Gene Mutation Important in Preconception and Pregnancy?
- 5 Common MTHFR Symptoms and How to Manage Your Gene Mutation
References
- Botto, L. D., & Yang, Q. (2000). 5, 10-Methylenetetrahydrofolate reductase gene variants and congenital anomalies: a HuGE review. American journal of epidemiology, 151(9), 862-877.
- Cristalli, C. P., Zannini, C., Comai, G., Baraldi, O., Cuna, V., Cappuccilli, M., … & La Manna, G. (2017). Methylenetetrahydrofolate reductase, MTHFR, polymorphisms and predisposition to different multifactorial disorders. Genes & Genomics, 1-11.
- De Mattia, E., & Toffoli, G. (2009). C677T and A1298C MTHFR polymorphisms, a challenge for antifolate and fluoropyrimidine-based therapy personalisation. European Journal of Cancer, 45(8), 1333-1351.
Folic Acid: What Happens When You Have Too Much?
What The UMFA?
UMFA stands for unmetabolized folic acid. If left unchecked, UMFA can cause a variety of health problems, but we will go over those later! The majority of folate found within the blood is 5-methyltetrahydrofolate (MTHF), which is not going to harm you; unlike UMFA! There is no need to worry about UMFA if you are getting all your folate from natural sources. However, if you are supplementing or eating a lot of foods fortified with folic acid, you may have a considerable amount of UMFA circulating through your blood!
What Are The Health Consequences Of Too Much Folic Acid And The Build Up Of UMFA?
Your body produces UMFA when it is unable to metabolize folic acid. The rate of folate metabolism is different for everyone, but generally, the body breaks down folic acid slower than folate from natural sources (4). Only people who are supplementing with or consuming folic acid fortified foods are at risk for having UMFA present in their blood. UMFA can cause a variety of problems within the body including (9,11):
- Impairment of the immune system
- Cancer grows more easily
- Lowers iron
- Slows cognition
- Impairs memory
Why UMFA Builds Up In The Body
Now that you are aware of the health consequences of UMFA, it’s important to know how and why UMFA begins to build up. The body metabolizes and moves folate around the body through several pathways (1,10):
- Tissue uptake and storage
- Secretion into bile
- Reabsorption
- Hepatic metabolism
- Renal excretion
If any of the listed folate pathways become saturated, the body will begin to build up UMFA within blood serum. Remember that the body has a harder time metabolizing synthetic folic acid compared to naturally occurring folates. See our article on “How Does Folic Acid Supplementing or Fortifying Affect Your Body?” for more information!
The majority of the burden to metabolize folic acid falls onto the liver; where the bulk of the DHFR enzyme (the enzyme responsible for metabolizing folates) is made. The liver has limited capacity to reduce folic acid into folate due to natural biological limitations. If you are supplementing >200ug of folic acid, it will lead to a temporary jump in unmetabolized folic acid in your blood serum (3,6).
3 Steps You Can Take To Reduce UMFA
There are three main ways to reduce folic acid and prevent the build-up of UMFA within your blood:
- Take folinic acid or 5-MTHF (For more information read our article A Guide to Folate Supplementation”)
- Avoid foods that are fortified with folic acid (fortified grain products)
- Eat more food that has naturally occurring folates (See our article on the top 20 folate containing foods https://www.mthfrsupport.com.au/top-20-folate-containing-foods/)
Take Aways
UMFA is only a problem if you are consuming folic acid on a regular basis. Eating some fortified foods every now and then is not going to kill you, but if you are having trouble becoming pregnant you need every advantage you can get. Reducing consumption of synthetic, and metabolically demanding, folic acid decreases the amount of UMFA found in blood serum — giving you a fertility advantage! 
References
- Aiso, K., Nagasue, M., Nozaki, T., Shimoda, M., & Kokue, E. (2001). Comparison of dihydrofolate reductase activities for folic acid in pigs and rats using in vivo and in vitro evaluation techniques. Journal of nutritional science and vitaminology, 47(2), 96-101. (4)
- Bailey, R. L., Mills, J. L., Yetley, E. A., Gahche, J. J., Pfeiffer, C. M., Dwyer, J. T., … & Picciano, M. F. (2010). Unmetabolized serum folic acid and its relation to folic acid intake from diet and supplements in a nationally representative sample of adults aged≥ 60 y in the United States. The American journal of clinical nutrition, 92(2), 383-389.
- Kelly, P., McPartlin, J., Goggins, M., Weir, D. G., & Scott, J. M. (1997). Unmetabolized folic acid in serum: acute studies in subjects consuming fortified food and supplements. The American journal of clinical nutrition, 65(6), 1790-1795.
- Mudryj, A. N., de Groh, M., Aukema, H. M., & Yu, N. (2016). Folate intakes from diet and supplements may place certain Canadians at risk for folic acid toxicity. British Journal of Nutrition, 116(7), 1236-1245.
- Obeid, R., Kirsch, S. H., Dilmann, S., Klein, C., Eckert, R., Geisel, J., & Herrmann, W. (2016). Folic acid causes higher prevalence of detectable unmetabolized folic acid in serum than B-complex: a randomized trial. European journal of nutrition, 55(3), 1021-1028.
- Patanwala, I., King, M. J., Barrett, D. A., Rose, J., Jackson, R., Hudson, M., … & Jones, D. E. (2014). Folic acid handling by the human gut: implications for food fortification and supplementation. The American journal of clinical nutrition, 100(2), 593-599.
- Pfeiffer, C. M., Sternberg, M. R., Fazili, Z., Lacher, D. A., Zhang, M., Johnson, C. L., … & Berry, R. J. (2015). Folate status and concentrations of serum folate forms in the US population: National Health and Nutrition Examination Survey 2011–2. British Journal of Nutrition, 113(12), 1965-1977.
- Pfeiffer, C. M., Sternberg, M. R., Fazili, Z., Yetley, E. A., Lacher, D. A., Bailey, R. L., & Johnson, C. L. (2015). Unmetabolized folic acid is detected in nearly all serum samples from US children, adolescents, and adults. The Journal of nutrition, jn-114.
- Plumptre, L., Masih, S. P., Ly, A., Aufreiter, S., Sohn, K. J., Croxford, R., … & Kim, Y. I. (2015). High concentrations of folate and unmetabolized folic acid in a cohort of pregnant Canadian women and umbilical cord blood. The American journal of clinical nutrition, 102(4), 848-857.
- Russell, R. M., Rosenberg, I. H., Wilson, P. D., Iber, F. L., Oaks, E. B., Giovetti, A. C., … & Press, A. W. (1983). Increased urinary excretion and prolonged turnover time of folic acid during ethanol ingestion. The American journal of clinical nutrition, 38(1), 64-70. (5)
- Sawaengsri, H., Wang, J., Reginaldo, C., Steluti, J., Wu, D., Meydani, S. N., … & Paul, L. (2016). High folic acid intake reduces natural killer cell cytotoxicity in aged mice. The Journal of nutritional biochemistry, 30, 102-107.